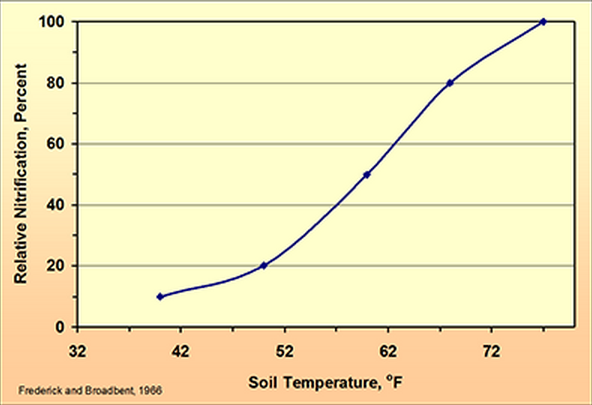
For that reason, a way to slow conversion of ammonium to nitrate is to have cold soil temperatures (example of soil temperature effect on nitrification shown in Figure 1). The optimum temperature for nitrification is around 90 F. Below 50 F the rate slows rapidly, but nitrification continues until 32 F. Soil temperature cannot be controlled, but because soils cool in the late fall, then nitrification of late-fall applications will be reduced. The later one waits to apply the better — soils with colder temperatures means less nitrification and a greater probability that soil temperature will not rebound to warm levels.If you are considering fall application of anhydrous ammonia to corn fields (the only nitrogen fertilizer recommended for fall application), remember to wait until soil temperatures cool to 50 F and continue to trend colder. The same guidance applies to manure with high ammonium nitrogen content, like liquid swine manure.
The form of nitrogen that can potentially be lost from soils due to wet conditions is nitrate (NO3-). The form applied as anhydrous ammonia is NH3, which converts to ammonium (NH4+) in water. Because ammonium is a positively charged ion, it is attracted by electrostatic forces to the negatively charged soil. Ammonium is not leached or lost by denitrification (conversion to nitrogen gas).
Therefore, it will stay in soil even if the soil becomes excessively wet. Nitrate, which is produced by soil microbes from ammonium in a process called nitrification, is a negatively charged ion and is repelled by the negatively charged soil, and is leachable and subject to denitrification.
Since nitrification is a microbial-mediated process, the rate is influenced by several factors that affect biological activity, such as ammonia in soil water (inhibits nitrification), temperature, soil aeration (only occurs in aerobic soils), soil pH range from 4.5 to 10.0 (optimum at pH 8.5), and soil moisture (highest at field capacity). But the largest influence is soil temperature.
| Figure 1. Effect of soil temperature on nitrate formation. |
|
Nitrification inhibitors slow the conversion of ammonium to nitrate. If more ammonium remains in soil during wet springtime periods, then less nitrate will be present and subject to loss. Nitrification inhibitors are not foolproof. They temporarily slow, but do not stop nitrification and formation of nitrate. They degrade in soil, which lessens effectiveness over time. Warm soils that speed nitrification also speed inhibitor breakdown, which means lower effectiveness and faster nitrification reestablishment.
Also, nitrification inhibitor impact on nitrogen loss is solely dependent on substantially more ammonium being present during excessively wet periods (typically occur in the springtime). If wet soils occur after the inhibitor looses its ability to effectively enhance ammonium remaining, then it will have no real impact.
Waiting for cold soils and/or using a nitrification inhibitor does not guarantee that fall-applied nitrogen will be a completely successful practice. Late fall may be warm, or warm and wet conditions may occur the next spring (a time period with historically high potential for wet soils and nitrate loss is May through June). However, if one decides to make applications in the fall, then waiting until soils are cold is better than applying early.
Remember, the decision to begin fall nitrogen application is not the first day that temperatures reach 50 F, rather, when the trend is for sustained soil temperatures below 50 F and continued cooling. The dates when soils cool below 50 F vary considerably, from late October to late November. Therefore, watch for temporary cold spells, especially early in the fall. Also observe the 6- to 10-day weather forecast, where a forecast for above average temperatures may signal soil warming.





