By Emerson Nafziger
Last fall, with funding provided through the Illinois Council for Best Management Practices (C-BMP), GROWMARK, C-BMP, and the University of Illinois initiated the N-Watch soil sampling program to see how much inorganic N remained in the soil following the drought of 2012.
|
|
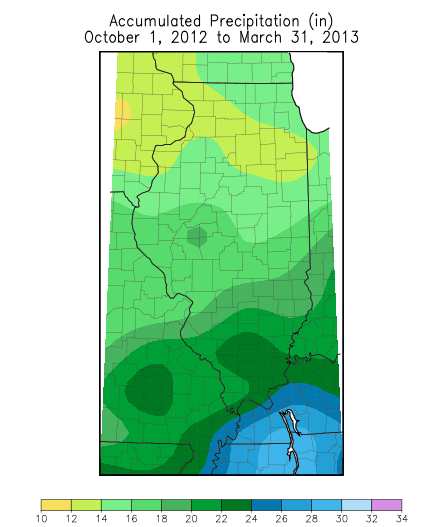
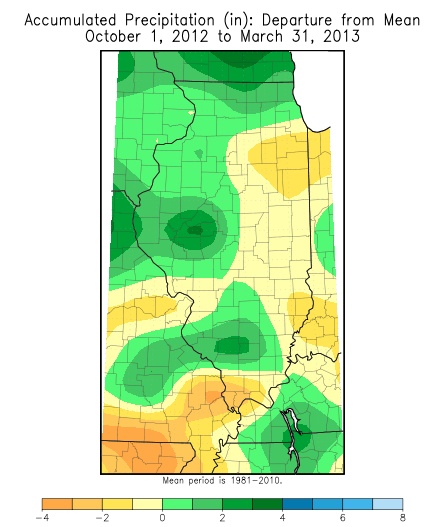
| (Figure 1. Precipitation and departure from normal (in inches) in Illinois, October 1, 2012 through March 31, 2013. Source: Midwest Regional Climate Center.) | |
Fall sampling revealed fairly high amounts of soil N, with 151 samples statewide averaging 19.5 ppm of nitrate-N in the top foot of soil. We multiply this time 4 to get lb of N per acre, so soils represented by these samples had an average of 78 lb of nitrate-N per acre.
Samples from northern Illinois had higher levels (26 ppm) than those from central and southern Illinois (both 18 ppm), even though 2012 corn yields in northern Illinois were higher than in central or southern Illinois. The second foot of soil depth had more than 15 ppm of nitrate-N, which meant another 62 lb of nitrate-N per acre, for a total of 140 lb N per acre.
One reason for fall sampling is to inventory soil N in order to know the potential for loss of leftover soil N. If the weather stays dry through the fall and winter, we expect minimal loss of soil N.
But precipitation returned to normal over most of Illinois during the past 6 months, most areas showing only small departures from normal over the October 2012 to March 2013 period (Figure 1). Normal precipitation from October 1 through March ranges from about 12 inches in the northern edge of Illinois to about 20 inches at the southern tip of the State.
While nitrate-N moves readily down into the soil profile as water moves down through the soil, the lack of rainfall during the 2012 growing season meant that nitrate from fertilizer N and from soil organic matter simply accumulated. Water only moves as far down as it takes to wet the soil, and in a very dry soil, it can take 6 to 10 inches or more of water to wet the soil. Even more water is needed to move through to deeper layers and into tile lines. Indications are that most tile lines in Illinois have been running for the past few weeks in the areas that were dry longer, and for the past couple of months in areas that received more rain and rain starting earlier.
Not surprisingly, a few reports in recent weeks from sampling tile line outflow show elevated levels of nitrate-N. This is normal for spring outflow, but with little or no tile line outflow as N accumulated last summer and fall, and with the large amounts of nitrate we found in fall sampling, the flush of nitrate-N may be larger than normal this spring.
How much soil N has been lost?
Dan Schaefer of C-BMP took both fall 2012 and spring (March) 2013 samples in a few fields in east central Illinois that can give us some estimates of N loss. In one field where N was applied in the early spring in 2012, October samples had about 16 and 13 ppm of nitrate-N in the top and second foot of depth. In mid-March 2013, those numbers had fallen to 6 and 10 ppm, respectively. Assuming no net conversion of N from ammonium to nitrate, this reflects a loss of about 50 lb of nitrate from the top two feet. Much of this probably remains in the soil below the top 2 feet, but some may have entered the drainage tile by now.
In a second field in which N was applied as spring sidedressed ammonia, nitrate-N went from 24 and 8 ppm in the first and second foot of soil last October, to 9 and 18 ppm, respectively, by mid-March of this year. That represents a net loss of only about 20 lb of N per acre, but a considerable amount of movement of nitrate from the top to the second foot. Nitrate losses have possibly been limited because it took so long (and so much precipitation) for dry soils to rewet to the point that water started to move through the profile.
Ammonium-N is immobile in soil, but in warm soils, microbes generally convert it rather quickly to nitrate-N. Because of this we normally see only 2 or 3 ppm of ammonium-N in either the fall or the spring. We found an average of about 5 ppm of ammonium-N in both the top and second foot of soil in the 2012 fall samples. In the second field described above, the fall sample had about 11 ppm of ammonium-N in the top foot. This could have come from spring-applied ammonia or from mineralization of soil organic matter after rain in September. The surprise is that the March sample still had 9 ppm of ammonium-N in the top foot. It is clear that there was limited net conversion of ammonium to nitrate in the top foot of soil between mid-October and mid-March.
Does this mean that ammonia applied in the fall of 2012 is still present? Most of the ammonia was applied after soil temperatures had dropped to below 50. Soils also stayed relatively cool, with average soil temperatures at the 4-inch depth of 49, 45, 34, and 34 degrees in November, December, January, and February, respectively. In contrast, 4-inch average soil temperatures in January and February were 39 and 41.5 degrees, respectively, in 2012. There were only 3 days in January and February 2013 with average 4-inch soil temperature of 40 degrees or more. So we think that soil temperatures have stayed low enough this winter to minimize the conversion of ammonium to nitrate.
We don’t have many direct measures of how much ammonia applied in the fall of 2012 is still present, but based on a few estimates, we believe that loss of NH3 applied in fall 2012 has been relatively small. One field that Dan Schaefer sampled in October 2012 had 11.5 and 7.6 ppm of nitrate-N, and 3.7 and 2 ppm of ammonium-N in the top and second foot, respectively. In November, 100 lb of N as NH3 was applied. March samples showed 6.5 and 11 ppm nitrate-N and 12.7 and 2.4 ppm of ammonium-N in the top and second foot, respectively. Probe samples from the application band are not a very reliable way to measure N, but if we assume a 35-lb loss of nitrate-N (average of the other two fields described above), the probe sample “found” about 2/3rds of the N that was applied last fall, and about half of that remains in the ammonium form. Compared to some fall-to-spring changes in ammonium-N reported in the literature, we think that nitrification (conversion of ammonium to nitrate) and loss of fall-applied N have been less than normal this winter.
Other sampling done over the past month, in some cases to see if winter wheat should have N topdressing rate adjusted, has shown nitrate-N to be fairly low, often less than 5 or 6 ppm. The winter wheat crop would not have taken up much N when samples were taken, but soil nitrate-N does drop by 1 ppm for each 4 lb. of N present in the wheat (or other) crop at the time of sampling.
Spring Sampling, 2013
The priority for spring sampling should be those fields where corn in 2013 will follow corn (from which fall samples were taken) in 2012. We suggested last fall that any spring sampling done in order to adjust rates for the 2013 corn crop should best be done close to corn planting time, or at sidedress time. However, it would be useful if at least some of those sites where fall soil N levels were high could be sampled within the next few weeks so that we could better guess how much additional sampling would be useful.
I suggest that sites with fall nitrate-N above 25 ppm nitrate-N in the top foot (that is about 25% of Illinois sites) be sampled first, but any and all sites can be re-sampled if volunteers are willing to do this. All those who coordinated sampling last fall and who requested forms and shipping materials through me will get an email in the next few days with brief instructions. The process will be the same as it was last fall, with samples sent to A&L for analysis, and funding provided through C-BMP. Those who sampled under GROWMARK’s direction last fall will continue in that program this spring.
Those who did not sample fields last fall but would like to do so this spring are also invited to participate. Instead of sending requests through me, however, we ask that those sampling this spring for the first time send requests to Jean Payne jeanp@ifca.com at the Illinois Fertilizer & Chemical Association, who will pass along the request for sampling materials and instructions.
It might be difficult to get the deep (1 to 2-ft) samples in fields where soils remain wet. We think the 1-ft sample will show us how much N rate adjustment might be appropriate. Those willing and able to take samples from the second foot are encouraged to do so, however.
It is best to take samples before any N has been applied in the spring, though we can avoid the band if, for example, some starter was used at planting and sampling is done after that. Combine in a bucket enough samples to represent the area you want to represent, and take a subsample of this to send to the lab for analysis. Soils should be sent for analysis as soon as possible, and kept refrigerated if needed, to minimize N transformations before analysis.
Adjusting N rates based on spring sampling
The pre-sidedress N test (PSNT) and the pre-plant N test (PPNT) were developed to make N rate adjustments based on N already present in the spring. Adjustments are not generally suggested if the soil has less than 10 ppm of nitrate-N in the top 6-7 inches (10 ppm is about 20 lb. of nitrate-N per acre), and no additional fertilizer N is suggested if the surface soil has more than 25 ppm nitrate-N (some states use 20 and some use 30 ppm as this limit). Fertilizer N rates are decreased as surface soil nitrate-N increases from 10 to 25 ppm.
Because of uncertainty in sampling, many universities suggest making adjustments as ranges; for example, the N application rate might be reduced by 30 to 50 lb. N per acre if the soil has 10 to 15 ppm of nitrate-N; by 60 to 120 lb. N if the soil has 15 to 20 ppm, etc. In practice, adjusting N rate based on nitrate-N present has often been most useful in fields where a lot of organic N – from manure or forage legumes were grown previously – was added, in which case it is a test for how much N mineralized. Spring soil N testing also requires sampling and then waiting for results during a busy time. But it can be used if we suspect that there are more than normal amounts of soil N. In cases where there has been wet weather and some N loss, it can also be used to help decide whether or not to make a supplemental N application.
While the University of Illinois has not actively promoted the use of the PPNT or PSNT, it is logical to apply less than full N rates if spring samples show an appreciable amount of soil N already present. We think that a reasonable way to do this is to calculate lb. nitrate-N per acre (ppm of nitrate-N in the top foot times 4) and to subtract this from the normal N rate. It may be safer not to make any adjustments if nitrate-N is less than 10 ppm, since we would consider low levels to be normal. But as an example, finding 20 ppm in a sample would suggest a reduction of 80 lb. N per acre in the fertilizer N rate. That’s conservative – 30 ppm would rule out any more N under the PSNT guidelines in use, but would mean lowering rate by 120 lb. based on what we’re proposing here.
Sampling uncertainty does mean some uncertainty in N rate adjustments. That’s a concern, especially if we sample only a small area in the field. So spring resampling in only the small area where fall samples were taken under the N-Watch program, while it meets the important objective of measuring changes from fall to spring, may not be adequate for making adjustments for a whole field. At minimum, samples taken for N rate adjustments should be taken from different soil types in a field to see if soil N levels have enough consistency to warrant adjustments.





